At Distill Health, we have the pleasure of engaging with an incredible variety of different medical devices and technologies. The MedTech sector is incredibly diverse, encompassing everything from ocular implants, to aesthetic laser platforms, to drug delivery straws — a remarkable range of innovations with an equally diverse set of marketing and communication challenges that we love to help them solve.
These days, though, medical devices and their innovators increasingly have one challenge in common. They turn to a medical device marketing agency like Distill Health because their market-building efforts have hit a wall. And more often than not, that wall is actually a door: to the C-Suite.
The corporate brass ring has been the Golden Fleece for most B2B marketers for a while now, but it remains a challenging target for many medical device brands. Many MedTech product owners remember the sainted days when a rep could walk into a practice or OR, demo the device to the clinician/surgeon/physician and his/her team, answer a few questions, and sell some units on the spot. Today, those same POs and reps can still visit the clinic or OR, but the real sell starts upstairs: with the COO in charge of procurement, the CMO in charge of clinical protocols, or the CFO who answers to the private investors who are now in charge.
Which is why more and more of our partners have the same burning question for our team: how does a promising medical device get through to the C-suite?
Here are a few of the key tips we always offer first.
Story first, tactics second
One catch: it’s preloaded with saline solution.
Like any smart marketing strategies, tactics that can reach the C-suite are only as effect as the content they deliver. So before you start planning the best ways to engage the C/I/E/M/T/O/O, before you spend the money to buy their clicks, think long and hard about what happens after that click. The first question shouldn’t be how to get their attention. It should be: what’s the story that will hold it?
No matter which C-suiter is your target — a critical first strategic decision in its own right — there’s no substitute for that story. It should have all the qualities of a high-value therapeutic agent: targeted, fast-acting, and all about value and outcomes. It should show how your medical device can cure specific practice ills, boost a system’s performance, or protect valuable profit margins. And more than anything, it should be 1000% right and ready before you invest in delivering it to any C-level target.
Lay the digital groundwork
Here it is: The C’s aren’t actually hard to reach.
After all, they Google like any other B2B marketing target. In fact, according to Forbes, executives consider online information sources to be the single most valuable ones available to them. They look for answers to pertinent practice questions and search for information relevant to their business. They may also have support staff who they task with sourcing and delivering that information to the C-suite.
That puts even the most inaccessible, over-scheduled executives within reach of a smart digital marketing plan. It also gives medical device brands a valuable opportunity to digitally build a foundation of awareness, credibility, and interest with their C-level audiences in advance, long before they set foot in the C-suite itself.
To secure that foundation in advance, though, brands need to invest: both in capturing their online audience’s attention, and in rewarding it with valuable, high-quality content.
Digital marketing tactics like SEM, SEO, and PPC are highly effective at driving online traffic, but achieve their best results through smart keyword strategy and optimization over time. And if the C-suite is your target, the site you drive to needs to be premium. According to a Quora study of 1300 global corporate leaders, 91% of C-level execs won’t give a brand a second chance if they’re unimpressed by their first experience on Brand.com. That means nine out of 10 times, your website gets exactly one shot to deliver your brand story — so both need to be powerful, swift, and compelling enough to catch a C’s interest in a single visit.
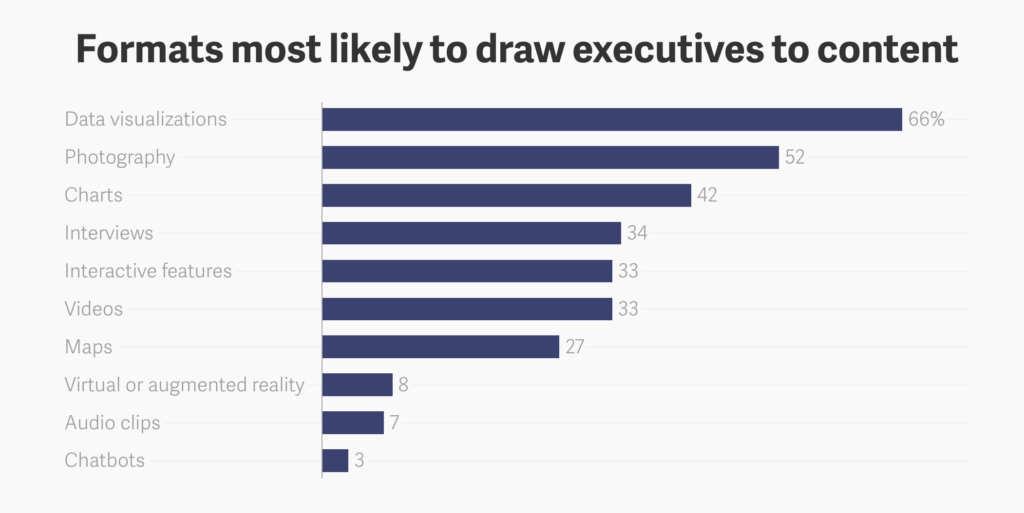
Show them something they don't know
Speaking of “show”: Make it visual. To sum it up, here’s exactly what C’s want to see in branded content:
Always make it personal
But don’t despair, hand-shakers. For all the power of a strong story delivered through smart digital marketing strategy, successfully marketing a medical device to the C-suite still takes diligent face-to-face, hands-on engagement with the people who fill the fanciest chairs in the office. It takes both effective digital awareness-building and personal relationship-building.
Balancing both those efforts is critical, and an effort we recommend to every medical device brand we service. Digital tactics can be incredibly effective, but healthcare professionals are awash in the same sea of online content as the rest of us. Win their digital attention, and the best way to reward it is often personal attention — to their specific business goals, practice challenges, and patient needs. Quality content can plant the seed of your product’s value in precisely the right ground at the right time. But to make sure that seed grows into a secure, productive relationship, always have someone prepared to water it.
